First in HumanInnovationPartnership First patient-derived in vitro model to study proteostasis deregulation in disease
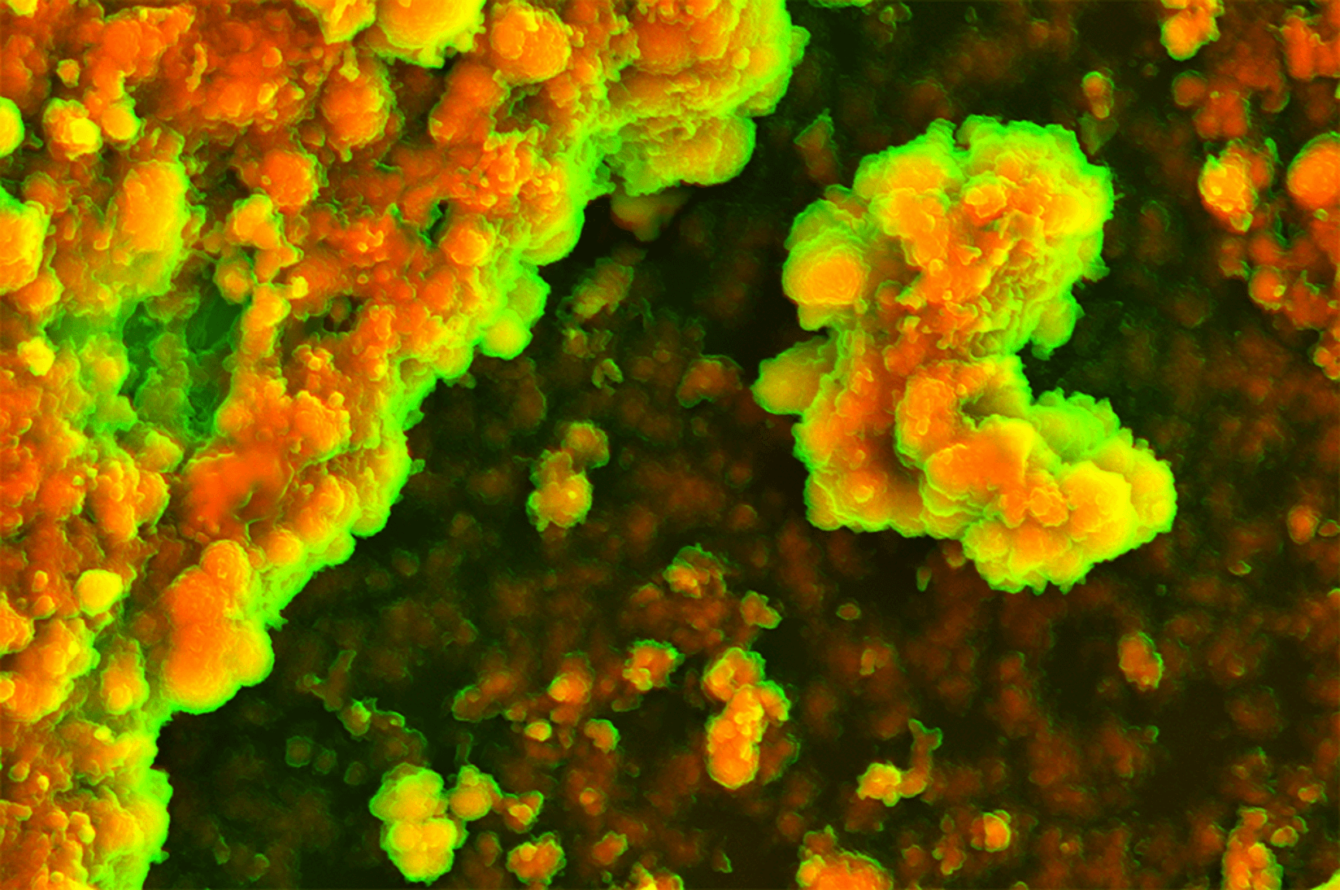
Innovative collaborative study established first in vitro model to study protein homeostasis (proteostasis) and effects of its disruption on the integrity of the surrounding tissue, with direct applications in biological, bioengineering and drug research.
Researchers from Imperial College London and King’s College London used human mesenchymal stromal/stem cells (hMSC) to establish a reliable in vitro model to study proteostasis perturbations. Using this model, they were able to investigate how pharmacological inhibitors could affect overall tissue integrity and other functional properties, in a quantifiable manner.
During differentiation of hMSC into bone structures, they are particularly dependent on mechanisms that control proteostasis, with enzyme VCP/p97 playing a key role in protein degradation and maintenance of skeletal and neurological health. Deregulation of VCP/p97 is associated with several neurodegenerative and age-related disorders, and drugs targeting VCP/p97 showed therapeutic potential against cancer and certain types of epilepsy. Researchers harnessed this dependence on proteostasis to manipulate the process with pharmacological inhibitors to varying degrees and quantify physical changes, such as the biochemical composition of the bone, as well as its stiffness stiffness and adhesiveness.
This first in vitro model was developed using hMSC from Imperial College Healthcare Tissue Bank, and demonstrated that VCP/p97 inhibition results in softer bone-like tissue, with less protein and more crystalline material, which could explain some pathologies associated with VCP/p97 deregulation, including cancer.
Sandra Loaiza, Head of Operations at the John Goldman Centre for Cellular Therapy at Imperial, and first author of the study, explained further: “This study is important as it is the first report of an in vitro model that can assess functional effects of proteostasis perturbations quantitively. This development will be beneficial for researchers studying these diseases and pharmaceutical companies developing therapies to treat them. We hope that our findings will ultimately lead to better treatment options for our patients.”
When addressing the potential applications of this model, senior author Dr Eileen Gentleman from the Centre for Craniofacial and Regenerative Biology at King’s College London, said: ‘Our findings have implications across many fields. For example, in the field of tissue engineering, researchers try to create living tissues in the lab to replace those that have been damaged or are diseased. They often do this by creating 3D structures, or scaffolds, using the matrix secreted by stem cells. However, our findings suggest that proteostasis perturbations can alter the composition and mechanical properties of scaffolds created by stem cells, rendering them different than the native tissue. Because of this, researchers may have to consider proteostasis as an important factor in making tissue engineering scaffolds.’
Dr Holger Auner, from the Centre for Haematology in Imperial College London, and senior author, commented: “Our findings have a wide range of possible implications for cancer therapies and reveal that treatments may have very unexpected effects on tissue.”
This study is an excellent example of convergent science, where bioengineering and science address clinically-relevant and -useful questions. Such multi-disciplinary approach is one of the strategic aims of the NIHR Imperial BRC Cancer Theme, where Dr Auner is a researcher and Dr Sandra Loaiza is a non-medical healthcare professional, whose PhD was directly supported through the Theme. The study was funded by NIHR Imperial BRC, Cancer Research UK, and support from Wellcome Trust and Leverhulme Trust.
This collaboration also recently demonstrated that when encapsulated within hyaluronic acid-based hydrogels, hMSC quickly modify their surroundings via protein secretion and/or matrix degradation, which ultimately impact their fate. The study was published in Nature Communications, you can read about it here.
Image Credit: Dr Holger Auner, Cancer Cell Protein Metabolism Group at Imperial College London