InnovationPrevention NIHR Imperial BRC Organoid Facility takes inventions a step closer to disease modelling and precision medicine

In a ground-breaking stride towards advancing medical research, earlier this year, the NIHR Imperial Biomedical Research Centre (BRC) proudly inaugurated the state-of-the-art Organoid Facility. This monumental achievement is the result of years of meticulous planning and the outstanding contributions of researchers like Tamir Rashid, Gary Frost, Nick Powell, and Harry Leitch, who laid the foundation for what promises to be a revolutionary venture in the field of organoid science. Generously backed by five years of strategic funding from the NIHR Imperial BRC, the Organoid Facility is poised to become a pivotal resource, not just within Imperial College but across the academic community.
Dr. Tamas Korcsmaros, Senior Lecturer in Intestinal Epithelial Biology for the Department of Metabolism, Digestion and Reproduction shares more about the Facility and how they hope to transform precision medicine.
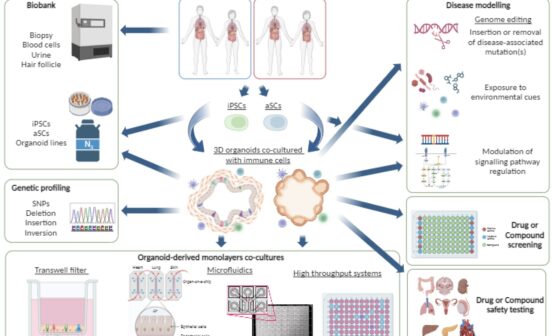
Understanding diseases better and improving how we model them has been the focus of biomedical research for the last couple of decades. Relevant, scalable and useful animal models have been established to understand the basic biology of cellular and mammalian systems in vivo – experiments conducted for research purposes on whole living organisms. Rodent models have been used for preclinical and toxicology testing of promising drug molecules. To complement these efforts, immortalised, cancer-related human cell lines have been used to understand in vitro (experiments conducted for research purposes outside of a living organism e.g., test tube) how these systems work in a human cell. However, all these systems possess different limitations: Animal models are often not completely resembling human processes, especially when we are thinking of host-microbe interactions for example, where a mouse gut is very different from a human gut. Human cell lines are often altered due to their immortalization. In addition, many cell types cannot be kept in cell lines, resulting in no data or testing option on key cell types. Around 15 years ago a new technology started to emerge based on isolating and differentiating specific stem cells of an organ. With this approach, a simplified but 3D models of an organ, mostly containing only its epithelial layer can be developed. They are called organoids.
With organoids, we can model key cell types or their dysfunction in specific diseases such as cystic fibrosis. We can also use organoids to test the effect of drug molecules on human patient-derived samples and check which chemotherapy agent would work best for the specific tumour of a patient. So, organoids have the potential to revolutionise how we model diseases and how we can develop patient-tailored therapies (i.e., precision medicine). Nonetheless, organoids are not perfect models (anyway, there are no such things, otherwise they are not models). Most organoid cultures only contain epithelial cells and often lack disease-relevant immune cells, for example. Also, it is still very challenging to grow organoids in a standardised way that allows large comparative studies of molecules or different patients. The added complexity of organoids over cell lines (more cell types, often 3D structure, sensitive culture to grow and differentiate) often increase the challenges to detect or monitor molecular or cellular changes with imaging or omics approaches. However, I believe at Imperial College London we have a very special situation, where we have the chance to address many of these challenges and take this relatively new biomedical model to the next, more translational stage.
It took many years to establish The BRC Organoid Facility and lots of planning and previous excellent work has been done by the groups of Tamir Rashid, Gary Frost and Harry Leitch to demonstrate what one can achieve with these models. Thanks to the five-year strategic funding from the NIHR, we were able to establish this facility to support organoid-related work College-wide. While other academic organisations often have a single laboratory for organoid work, or even a facility for specific organ-model organoids, at Imperial, we now have a Facility that aims to cover many models by the end of this five-year funding round and also support the training of interested colleagues. By establishing this community and training hub, the Imperial Organoid Facility will lower the barrier to access these new models and facilitate how we model diseases and develop precision medicine approaches.
There are many technological challenges when working with organoids: what materials we are using to provide the scaffold or a growing environment for the organoids, how we change the media around them and how we provide a natural flow which can be achieved by different nanofabrication and microfluidic systems. Imperial is a special place as there is a vast expertise of these technologies in Imperial’s Faculty of Engineering. Thus, I am super-excited to work with engineering colleagues and complement their resources and interests with our needs to develop scalable and better organoid models.
The other limiting factor for an organoid facility is often the sourcing of the samples. This is another area where I am very optimistic. With the established Imperial BRC Tissue Bank and the integrated Imperial College NHS Healthcare Hospitals at St Mary’s, Chairing Cross, and Hammersmith, we can access patient samples and metadata through the biobank and generate organoid lines. We are aiming to develop organoid biobanks so people can access different patient samples or different organoids on a scale that is not really available in other organisations.
When we have lots of organoids in a biobank, and great methodologies to grow them, the most important question is how we can analyse them. And this is where the location of the Organoid Facility at Hammersmith is very relevant. With the close proximity of the National Phenome Centre and the Imperial BRC Genomic Facility, we have access to all the major omics platforms (mass-spec, sequencing, spatial imaging, etc). I am excited to work with colleagues from all these facilities to establish cross-facility pipelines with multi-omics analysis of organoid-based systems.
Overall, I’m very enthusiastic about organoid models. They are not just modelling human diseases, they can model diseases of a single patient too. It means, as a precision medicine tool, we can learn about the disease of that particular patient, and in the future propose tailored therapies based on these patient-derived organoid models.